About Us
Medical Science and Engineering Research (MEDSER) center is established at Indian Institute of Science Education and Research (IISER) Bhopal to bolster the multi-disciplinary and inter-disciplinary research in order to promote medical and healthcare innovations and translational research at the Institute. The center will be equipped with the state-of-the-art instrumentation to conduct research and innovations for diagnostic, therapeutic, and related healthcare purposes. IISER Bhopal has ten major departments viz. Biological Sciences, Chemistry, Earth and Environmental Sciences, Engineering Sciences, Mathematics, Physics, Economics Sciences, Data Science and Engineering, Humanities and Social Sciences and Chemical Engineering and three academic streams (natural sciences, engineering sciences, and humanities and social sciences). MEDSER will integrate the latest areas of research with the medical science engineering. These areas include smart devices and sensors for medical diagnose applications, machine learning/artificial intelligence/internet of things (IoT) for healthcare, cancer biology research, disease and diagnosis markers for early diagnostics and visualisation, novel material synthesis for healthcare. In addition, the center will also conduct outreach activities (symposium, workshops, seminars, meetings, etc) and training programs for the students.
Research
Smart devices and sensors for medical diagnose applications
Portable medical diagnostic devices are increasingly becoming an essential part of the healthcare system. The same has been evident specially during the Covid-19 pandemic for monitoring the chronic medical conditions. The mobile devices provide a personalized healthcare and on-site testing and data collection facilitating the computational analysis using machine learning/deep learning. Mobile devices also reduce patient’s exposure. Some of the applications that are in-use through mobile devices are measuring blood pressure through smartwatch for patients of hypertension, personal glucometers for diabetes management. Likewise, mobile applications for monitoring personal health are increasingly becoming popular.
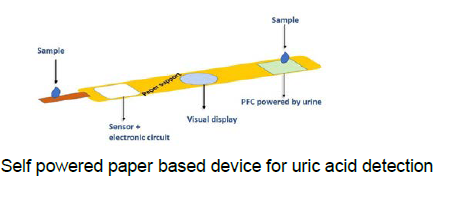
Faculties at the department of chemistry, biology, chemical engineering, and electrical engineering and computer science have the expertise in this area. Major areas of interest include development of novel microfluidic devices for applications in high throughput diagnostics, development of affordable semen analyzers for veterinary and human assisted reproductive technology development purpose, developing functional nanocomposite materials either in the form of films, fibres, or hydrogels, development of point of care diagnostic devices for medical and healthcare applications, bioelectronics, sensors and systems, wearable/printed/flexible electronics, microfluidic electronics, energy harvesting, and spintronics, smart healthcare and smart buildings. Other area of research interest is development of multi-organ therasonic devices, development of smart band for vital health parameter monitoring during pregnancy in low resource conditions, research on futuristic biomedical applications and bioelectronics, development of implantable electronics, electronic therapeutic monitoring systems, neuromorphic devices, advanced healthcare components, devices for the detection of IgA protein, development of electrical detection technique for SARS-Cov-2 specific antibodies using high electron mobility transistor-based devices for accurate detection, detection of HbA1C using portable electronic device, thermal cycler for PCR application, development of signal conditioning circuits with flexible electronics for wearable devices, smart self-powered paper-based analytical device for sensing glucose in blood and uric acid in urine/blood.
Machine learning/artificial intelligence for healthcare
Artificial intelligence (AI) in healthcare is typically the use of software and complex machine learning algorithms with self-learning neural networks to gather, process, interpret, and present medical and healthcare data mimicking human cognition. AI has been helpful in the areas of drug development and interactions, electronic health records, disease diagnosis, primary care, telemedicine, radiology, dermatology etc. Faculties from the department of chemistry, data science and engineering, electrical engineering and computer science are interested to take-up research under machine learning/artificial intelligence area. Some of the research problems relevant to the area are as under:
a. Biased signalling in G-protein-coupled receptors:
GPCRs are primary targets for more than one-third of the marketed drugs. However, drugs are generally equally potent in activating both the G-protein and b-arrestin pathways, thereby unable to separate therapeutic effects from adverse side effects. Over the past decades, though the ligand-dependent differential efficacy for distinct intracellular responses through the same receptor has been well-documented for many GPCRs, but for the most part both structural as well as dynamical basis for this selectivity are poorly understood. Thus, obtaining a detailed insight into the molecular signatures of biased signalling is therefore essential to design and develop novel therapeutic molecules for the effective clinical treatment of various diseases. Systematic investigation of the specific changes in the receptor domains that are caused by biased ligands and/or mutants, and the differences in the manner in which they elicit the underlying activation of that receptor is a key priority.
b. Thermodynamics and kinetics of β-arrestin activation:
β-arrestins are one of the major intracellular binding partners of GPCRs that bind in a phosphorylation6 dependent manner to the receptor. GPCRs activate different pathways via β- arrestins depending on the patterns of phosphorylation present at the C-terminus of the receptor. Despite the inevitable role in almost all physiological processes, the structural dynamics of β-arrestins and the concomitant activation mechanism are poorly understood. Focus is on understanding the sequence of events in β- arrestin activation along with constructing the kinetic transition networks between different metastable conformational states in its activation pathway. Studying how each phosphorylation barcode changes β-arrestin conformation by which it selects distinct downstream signaling pathway is a key interest.
c. Membrane-assisted aggregation of a-synuclein implicated in Parkinson’s disease:
The aberrant misfolding and aggregation of an intrinsically disordered neuronal protein called α- syn is a crucial step in the pathogenesis of Parkinson’s disease (PD). Understanding the aggregation mechanism of a-syn and the role of lipid membrane in this process is crucial as the role of lipid membrane is established in a-syn function vs. dysfunction, but the mechanism of toxicity in PD is yet undefined. In most of the cases the cause of illness is sporadic. There is no drug currently available that can either prevent or slow down PD. Clues might come from understanding the molecular details of membrane-assisted conformation and oligomerization of α-syn, which is a prime focus. Currently this knowledge is limited, mainly because the oligomers of α-syn are highly structurally diverse and exist as ensembles of various conformations. PD, like other neurodegenerative disease, affects mostly elderly populations, where change in normal lipid profile and enhanced oxidative stress commonly occurs with ageing. Studying the effects of alteration in lipid compositions (e.g., increased cholesterol level) and oxidative damages on the conformation change and aggregation propensity of a-syn is an emerging area of research. d. Inhibition mechanism of potent α-synuclein aggregation inhibitors: Since α-synuclein aggregates, especially pre-fibrillar oligomers, play a crucial role in PD, it is essential to have a molecular level understanding on the mechanism of aggregation of soluble α-syn monomers into oligomers. The primary objectives of computational research are: (i) identifying druggable hotspots where potent inhibitors can interact and bind, and (ii) explore the mechanism of their action in the prevention of α-syn oligomerization/fibrillation. e. Mining Clinical Texts to Improve the Quality of Treatment Plans and Biomedical Research using Natural Language Processing: Clinical texts also called clinical notes e.g., discharge summary, prescriptions, radiology reports etc. contain family 7 history, lifestyle, diagnoses, medications, treatment plans and various other medical information of the patients. Clinical notes are increasingly being used all over the world, but represent a vast, underused resource for biomedical research. The clinical notes are very subjective to the doctors, or the nurses writing them and lack a common structure or framework like the structured electronic health records. These notes also have grammatical errors, short phrases, abbreviations, local dialects, and misspelled words. To identify potential information, considerable data processing needs to be conducted on these clinical notes, such as spelling correction, word sense disambiguation, contextual feature extraction, extraction of diagnosis codes etc. The advances in Natural Language Processing (NLP) techniques have made it possible to explore the unstructured clinical text in a sophisticated way. There are many applications of NLP for knowledge discovery in clinical notes to improve the quality of treatment plans and biomedical research. NLP can investigate the association between drugs and possible adverse events, correlations between diseases (comorbidities) and it can be used for early prediction of signs of different diseases using the clinical notes. Furthermore, NLP can identify, analyse, and explore biomarkers for multiple diseases from the clinical texts. A biomarker is defined as a set of measurable quantities that can serve as an indicator of a patient’s health e.g., high cholesterol is a biomarker for being at risk of cardiovascular diseases. However, a broad range of ethical, legal, and technical reasons currently hinder the systematic deposition of these clinical texts and their mining. Hence, NLP techniques can also be used to de-identify the protected information (e.g., patient’s name, age, sex etc.) present in clinical texts to make the data available for further research. There are many other uses of NLP for information extraction in clinical texts. f. Systems biology, biomedical image processing and creation of microfluidics-based diagnostics devices through theoretical modeling, computational simulations and experimental studies. Cellular level model of the interaction of the immune response cells with the virus with special focus on explaining the long lived COVID-19 viral load data. AI in healthcare scheme to augment the image classification of malarial red blood cells, employing machine learning techniques to large scale biological networks and image data to predict new cellular and genetic functionalities and features from it. g. Dual-emissive ratiometric O2 probes for hypoxia and hyperoxia aimed toward bioimaging, biosensing, and disease diagnosis: Quantitative detection of molecular oxygen in biological environments has drawn significant attention using 8 ratiometric O2 probes. Any changes of oxygen concentration from a certain range in organs or tissues cause hypoxia and hyperoxia, which is related to many diseases, including solid tumours, inflammatory diseases, and cardiac ischemia etc. Unlike the organic fluorescent dyes, phosphorescent transition-metal complexes are known to exhibit reversible O2-responsive emission change because of energy transfer between the triplet metal complex-based emissive state and triplet O2 ground state. The development of dual-emissive ratiometric O2 probes allows more accurate O2 quantification. In the case of a dual-emissive fluorophore, one emitter is insensitive towards O2, which acts as an internal standard, while another phosphorescent emitter is sensitive towards O2, and the changes of emission intensities is monitored with respect to the reference fluorophore.
Internet of Things (IoT) for healthcare
The remote monitoring of patients has been made possible through Internet of Things (IoT). IoT has empowered medical practitioners to perform superlative care and proactively connect with their patients. The overall logic is to interconnect various devices with sensors, monitors, detectors, cameras, for data collection followed by the digitization of collected data and advanced analysis. This results in better healthcare, reduced treatment costs, improved process and performance for healthcare providers. Faculties from the department of electrical engineering and computer sciences and data science and engineering are interested to conduct research in this area.
Some of the research interests are development of IoT and IT based healthcare solutions, bioelectronic interfaces, and futuristic devices. a. Clinical question answering and dialogue generation: Currently, most of the tasks and enquiries at the hospital reception are resolved by the humans. This limits the task resolving rate and increases the operational cost. With the help of a chatbot model, a digital platform can be developed to resolve the tasks and queries without human intervention. A neural model can be trained on a publicly available english medical conversations dataset to resolve relatively simpler tasks. Its capabilities can be improved to handle complex tasks by further training the model on the recorded actions of the hospital staff. Use of contextual knowledge bases with transformer models to build the model. This will make the model better and 9 efficient over time. Various pre-trained medical language models like Clinical-BERT, Bio-BERT, etc. can be used to initialize the weights of the query encoder. Over time, with the help of feedback from the doctors, the system can be improved for better performance.
Cancer biology research
Research on the understanding the mechanisms behind cell growth, transformation of normal cells into cancer cells, and study on spread of cancer cells fall under cancer biology research. Department of biology and chemistry are interested in this research area. Some of the priority research themes are study on cancer epigenetics and alternative splicing in breast and head & neck cancer, understanding the nuclear pore biology and posttranslational modification induced physiological changes leading to enhanced cellular proliferation and cancer, to understand the connection between nuclear pore proteins' overexpression and the progression/ aggressiveness of cancer, to extend nuclear pore-related studies to hematologic and bone disorders ensued by the malfunctioning of constituent cells of these tissues, proteomic study on the analysis of SUMO modification dynamics during apoptosis and autophagy.
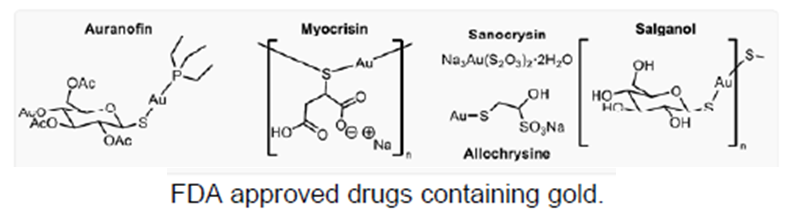
Material synthesis for anti-cancer potential: FDA approved drugs containing gold, namely Auranofin, Solganol, Myocrisin, Sanochrysin and Allocrysine, have witnessed enormous success in treating rheumatoid arthritis due to their immunosuppressive properties. Recently, gold complexes have also found tremendous potential as anticancer agents and some are under clinical trials at various stages. Gold-based organic molecules are potential alternatives to the largely used platinum-based anticancer drugs. Mechanistically, unlike platinum complexes (for instance, cisplatin) that target DNA, gold complexes exhibit a high propensity to target enzymes, especially those containing thiols, selenothiol, etc. This distinct targeting feature is attributed to the strong binding affinity of gold towards thiol (soft ligands) containing enzymes, such as TrxR, glutathione reductase (GR), and cysteine protease, which are overexpressed in cancer cells. Notably, this complementarity between the anticancer mechanisms of gold- and platinum-based complexes should also allow the former to be efficient for the treatment of patients who developed resistance towards platinum-based drugs. So far, a relatively large number of gold(I) complexes have been synthesized and evaluated for their anticancer activities in the literature. Breast cancer cells 10 However, reports on the consolidated study that aims at the rational design of gold complexes and thorough evaluation of their anticancer properties remained scarce. There is a need to rationally design various gold complexes bearing diverse ligands and investigate their anti-cancer potential.
Disease and diagnosis markers for early diagnostics and visualization
Diagnosis markers are the biological parameters that help in disease diagnose. Some of the key examples are presence of genetic variant and the levels of certain proteins in body fluids. Department of biology, chemistry, data science and engineering are interested to pursue research on disease and diagnose markers. Some of the key problems of interest under disease and diagnosis markers are:
a. Biology of epigenetics and development of diagnosis markers for liver and poultry diseases:
The upcoming area of interest is to decipher the role of epigenetics in tissue specific gene expression more specifically in poultry liver during development and infections and to develop certain markers for the diagnosis of diseases for the management of poultry health. Through the utilization of budding yeast as a model organism, identifying the significance of epigenetics and conserved cellular homeostatic pathways in response to promising anti-cancer molecules, metals and environmental pollutants is a research priority.
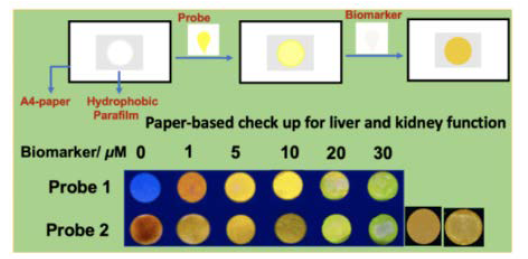
b. Rationally designed small-molecule probe for liver and kidney damage:
Accurate analysis of liver and kidney damage monitoring-biomarker is urgently required for chronically ill patients in a ‘point-of-care’ fashion. Lately, the number of kidney disease is exponentially increasing due to our fast lifestyle and food habit. The technology demanding ‘strip-based’ detection by exploiting the high sensitivity in solution as well as in solid-state has been determined. It provided a convenient and cost-effective analysis of the protein-based biomarker associated with kidney and liver damage. The abnormal concentration of the marker in body fluids can be a potential biomarker for liver and kidney malfunction.
c. Visualisation of sub-cellular process:
The recent advancement of fluorescence microscopy allows to investigate the cellular processes with high spatial and temporal resolution in all three dimensions. The discovery of genetically encoded fluorescent proteins (FPs) allows for endogenous labelling and makes almost any protein or peptide fluorescently visible inside live-cells. However, in many cases the poor photo-stability, variable maturation time of the chromophoric unit in FPs and bulkiness limit their applicability. Therefore, there is high demand for the development of novel fluoroprobe with desirable properties. Development of biocompatible fluorescent dyes for deciphering the inter-organelle communication with high resolution is the key priority.
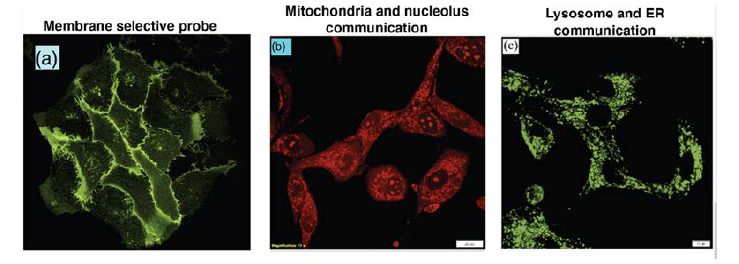
d. Fluorescent biomarking for live-cell imaging:
Live-cell imaging of subcellular organelles such as mitochondria, lysosomes, endoplasmic reticulum etc. has drawn a great attraction to visualize various biological phenomena. Fluorescence imaging has an advantage over other technologies (radioisotope labelling, electrochemical detections, MRI) in terms of sensitivity, safe-detection, tuneable fluorescence signal etc. A superior bioimaging fluorescent probe should have (i) high photostability for long-time bioimaging of organelles, (ii) sufficiently high solubility in subcellular medium, and (iii) low toxicity and biocompatibility. Fluorescent heteroaromatic compounds would be ideal candidates for bioimaging molecules. Since a limited number of bioimaging agents are available, current research is highly intensified to develop bioimaging molecules cost-effectively.
e. Strategies for proliferation, separation, and imaging of pancreatic β cells:
Currently 425 million people worldwide are diagnosed with diabetes. Out of these ~95% suffer from type 2 diabetes and ~5% suffer from type 1 diabetes. According to world diabetic federation, in South East Asia, especially in Indian subcontinents, ~82 million people are diagnosed with diabetes. In next 20 years, it is expected to increase to ~151 million. Apparently, the death and dysfunction of insulin-producing β-cell in the pancreas is the key pathological event in diabetes. As an alternative to the whole islet transplantation to the diabetic patient, iPS cell derived β-cells formation is expected to be promising for the treatment of diabetic patients. One of the pioneers in iPS cell derived β-cell generation is Prof. Dough Melton at Harvard Department of Stem Cell and Regenerative Biology. They have successfully stabilized the protocol by sequential modification of different signalling pathways and generated millions of β-cells from human pluripotent stem cell. They have validated the formation of β- cell by comparing the gene expression pattern between the iPS cell derived β- cell and pancreatic β-cell from a donor. However, the major bottleneck of this process is the overall yield of β-cell production - the typical yield of β-cell formation is 1-5%. Furthermore, this method also produces other undesired endocrine cells and contains undifferentiated iPS cells which could be tumorigenic. There is no available method to isolate these β-cells from the other pancreatic cell types. Research interest is to develop various chemical tools to separate and purify the β cells which are derived from iPS cells. Further interest is developing chemical tools to increase the yield by selectively delivering proliferating molecules and developing fluorescent probes for β cell imaging.
Novel material synthesis for healthcare
a. Soft nanomaterials for biomedical applications. These include hydrogels for drug delivery, polymers as
novel antimicrobials, underwater adhesives for biomedical applications, novel fluorophores for in cellulo
imaging applications, ROS generating metal-peptide complexes in anticancer applications.
b. Functional nanocomposites: developing functional materials in the form of films, fibres, or
hydrogels, hydrogel patches for transdermal drug delivery, smart hydrogels for wound dressing using
photothermal therapy, hydrogel-based scaffolding polymeric biomaterials for tissue engineering,
antimicrobial hydrogels for potential medical applications, injectable and biocompatible hydrogel for drug
delivery and tissue engineering, smart materials for wearable devices and flexible bioelectronics.
Goals
The Medical Science and Engineering Research (MEDSER) Center has been proposed to nourish a culture of medical and healthcare innovation at the Institute. MEDSER is proposed to interconnect the medical sciences with design and problem-solving skills of engineering. This includes:
Innovation in medical devices, medical imaging, computation, big data, regenerative medicine, drug delivery, technology transfer, and entrepreneurship.
The center would focus on the development of technology, products, and knowledge resource through interdisciplinary research and education efforts.
The center would aim to build strategic partnership with healthcare institutions, hospitals, and similar organizations to confront major medical challenges.
People

Prof. Sarman Singh
Mentor MEDSER, Adjunct Faculty, IISER BhopalStaff

Ms. Dimple Rajput
Project Office Assistant,MEDSER, IISER Bhopal
Mr. Aman Sharma
Project Research Assistant,MEDSER, IISER Bhopal
Mr. Rajesh Karn Kshatriya
MTS, MEDSER, IISER BhopalNews
MEDSER is organizing a 2-day workshop from June 30th-July 1st 2022 at IISER Bhopal campus
The MEDSER center was formally inaugurated on July 1, 2022by Prof. Upendra Kumar Singh, Director General – DRDO, New Delhi.The Indian Institute of Science Education and Research (IISER) Bhopal will be the first IISER Out of 7 sisters to formally establish a unique facility. This will be called Medical Science and Engineering Research (MEDSER) Centre. This centre has been established at IISER Bhopal to Nurture the togetherness of join efforts of doctors and basic scientists. The Director of IISER Bhopal, Prof. Shiva Umapathy told that this centre aims to encourage innovations done by its basic scientists that are applied to medical and healthcare needs including the diseases diagnostics, drug targets, artificial intelligence and machine learning. The first medical doctor appointed by the IISR Bhopal to mentor this centre is Prof. Sarman Singh, former director of AIIMS, Bhopal. Dr. Umapathy told that even though the centre was approved about 6 months ago, a formal inauguration is being done by Prof. Upendra Kumar Singh, Director General – DRDO, New Delhi.During the inauguration Prof. Upendra Singh applauded the efforts of IISER Bhopal for its new initiative and wished that many innovations will be made by the faculty of IISER Bhopal, in collaboration with AIIMS Bhopal and other medical institutes. He also emphasized that innovation in defense science should also be undertaken by this centre.Beside the chief guest Prof. Upendra Singh, and Prof. Umapathy, Dean Research Dr. Sanjeev Shukla and Prof. Sarman Singh also addressed the gathering. Prof. Sarman Singh shared his success stories in the field of innovation while at AIIMS in the field of visceral leishmaniasis and tuberculosis and encouraged medical faculty of AIIMS and other medical institutions of the central India to join hands with MEDSER centre to serve the society. He appreciated presence of various faculty members of AIIMS Bhopal as speakers or chairpersons and extending support for this collaborative initiative between two institutes.

Workshop
Following the inauguration, the MEDSER centre had a two days’ workshop on 01 and 02 July,2022
The workshop had lectures as well as discussion sessions from the Directors of various DRDO Laboratories, as well as experts from various institutions in communicable diseases, non-Communicable diseases, biomedical technologies, AI, and next-generation therapeutics.Various speakers in the workshop were: Dr. Manmohan Parida, DRDE, Gwalior; Dr. Jayanta Bhattacharya, Director International Vaccine Initiatives and THSTI, Faridabad; Dr. Anuj Sharma, world health Organization; Prof. Debasis Biswas, AIIMS Bhopal, Prof. Bhavana Sharma, AIIMS, Bhopal; Prof. Rehan Ul-Haq of AIIMS, Bhopal; Prof. Vaishali Waindeskar,Dr. Saurabh Saigal, Dr. Abhishek Goyal, Dr. Girish Bhatt and Dr. Neha Arya all from AIIMS,Bhopal; Dr. Ajit Chande, coordinator, MEDSER Centre, IISER, Bhopal; Prof. Chandra Verma,Singapore; Dr. Rati Sharma, Dr. Tanmay Basu, and Dr. Vishal Rai, Dr. Shantanu Talukdar, Dr.Sweta Lal, Dr. Mitradeep Bhattacharjee all from IISER, Bhopal; Prof. Shantanu Sengupta of IGIB, New Delhi; Dr. Anil Kumar Mishra, tor INMAS, New Delhi; Prof. Sanjeev Galande, Shiv Nadar University; Prof. Kumar Somasundaram, IISC, Bangalore; Dr. SharmilaBapat, National Centre for Cell Science, Pune; Prof. Jyotsna Shrivastav, Department of Paediatrics, GMC, Bhopal; Prof. Sankararamakrishnan, IIT, Kanpur; Dr. Debojyoti Chakraborty, IGIB, New Delhi; and Dr. Albeena Nisar of Tata Memorial Centre, Mumbai.The scientists also had group discussions to develop collaborative research projects and how better this centre can cater to the needs of the society.

Talk as on 24 June,2022

Dr. Rishikesh Pandey as a guest Speaker
Title -Morpho-Molecular Microscopy: A Label-free Sensing Platform for Cancer Diagnosis.
Abstract-Tumor genesis involves alterations in morphological, molecular, and genetic indicators of the cell as it transitions from normal to malignant. Fluorescence imaging has made enormous progress in single-cell Investigations, but its use is plagued by the need for a fluorescence probe and concern over photo bleaching. Label-free modalities such as Raman spectroscopy, on the other hand, offer optical phenotyping of a live cell without necessitating any exogenous contrast agent. However, slow speed and poor sensitivity limits clinical utility of spontaneous Raman imaging. We have developed a strategy called morpho-molecular microscopy (3M) that allows simultaneous measurements of complementary morphological and biochemical attributes. Our 3M system draws on the combined strength of quantitative phase imaging - to probe the morphological features- with Raman microscopy that provides molecular fingerprinting characteristics. This talk will discuss the use of the 3M system in live-cell imaging and its applications in immunology and leukemia diagnosis. The other focal point of this talk will highlight our clinical translational efforts in intraoperative tissue and it made it easier for students to understand. Dr. RishiKesh Pandey gave a very good speech about cancer Diagnosis. Student really enjoyed this talk; Dr. Pandey explained everything in detail..
Talk as on 16 Sep,2022

Prof. Hari Shankar Sharma as a guest Speaker
Title -Stunted Angiogenesis and Vascular Remodeling in the Failing Heart.
Abstract-Right ventricular hypertrophy (RVH) associated with impaired myocardial oxygen and nutrient supply are common features in the cyanotic congenital heart disease, Tetralogy of Fallot (ToF). Consequently, the right ventricle may manifest in altered molecular phenotype with a number of adaptive and inherited gene profiles which are largely unknown. The aim of the present study was to investigate the myocardial differential gene expression profile and to assess myocardial vascularisation in patients with ToF. DNA microarray analysis on right ventricular biopsies from ToF-patients operated for primary corrective surgery (referred as ToF-1; n = 12, mean age 0.5 year) and age matched controls (n = 6) was validated by Northern hybridisation and RT-PCR. Employing immunohistochemistry and video image analysis expression of vascular endothelial growth factor (VEGF), vascular density (by α-SMA and CD31 staining) and myocyte cross sectional area (Gomori's reticuline staining) were assessed in ToF-1 and adult patients (referred as ToF-2, n = 12, mean age 30 years) who underwent surgery for pulmonary regurgitation and compared the data with respective age matched controls (n = 6/12). DNA microarray analysis revealed altered expression pattern for 236 genes including enhanced (1.5-2.2-fold) expression of angiogenic factors and their receptors including; VEGF, flt-1, flk-1 angiopoietin-2, FGF-2, FGF-R1, PDGF-A, whereas, flt-4, Tie, TGF-β, TGF-β3R showed decreased (1.6-3.4-fold) expression in ToF-patients. Northern blot analysis verified the expression patterns of VEGF and flk-1 in both ToF-1 and ToF-2 patients. VEGF staining in cardiomyocytes was increased in ToF-1 (1.5-fold, p < 0.05) as compared to ToF-2. Video image analysis revealed enhanced vascular density (p < 0.01) with enlarged myocyte cross sectional area (p < 0.01), but vascular wall thickness remained unchanged in ToF-1 patients as compared to age matched controls. Our data suggest that RVH is associated with profound changes in gene profile for a number of genes, where VEGF/VEGF-R system contributes to enhance, but stunted myocardial angiogenesis in patients with ToF

Talk as on 13 March,2023

Dr.Umakant Sharma as a guest Speaker
Dr. Umakant Sharma, Ph.D, Assistant ProfessorDepartment of Surgery, University of Miami, Miller School of Medicine, USA were invited for a talk at IISER Bhopal. He delivered his lecture on 13 March, 2023 at Seminar Hall No 110, Visitors Hostel.
Title -Synbiotics Therapy: A Promising Strategy for IBD in the Context of Prescription Opioid.
Abstract-Opioids are the most prescribed analgesics for pain management in Inflammatory Bowel Diseases (IBD). Opioid use has been linked with increased complications, hospitalizations, decreased quality of life, and mortality in IBD patients. However, the consequences of opioid use on IBD severity are not well defined. This is the first study investigating consequences of prescription opioid hydromorphone on clinical manifestations of the disease in IBD patients and both dextran sodium sulfate (DSS)-induced colitis and spontaneous colitis mouse model (IL-10-/-) of IBD. We demonstrated that hydromorphone used for pain management in IBD accelerates progression of disease by dysregulation of the gut microbiota, leading to expansion of pathogenic bacteria, translocation of intestinal bacteria, immune deregulation, and sustained inflammation. Predictive metagenomic analysis of the gut microbiota revealed high abundance in the bacterial communities associated with virulence, antibiotic resistance, toxin production, and inflammatory properties. Our results showed that hydromorphone modulates tight junction organization in a myosin light chain kinase (MLCK)-dependent manner and treatment with ML-7, a MLCK inhibitor, ameliorates the detrimental effects of hydromorphone on DSS induced colitis. Current IBD therapies focus on antibiotic treatment and suppressing immune responses. In this study we hypothesized that “the restoration of gut homeostasis by modulating the gut microbiota with the use of synbiotic could be a relevant therapeutic strategy for IBD.” A synbiotic, a mixture of a probiotic and prebiotic, has the advantages of both microbiological and nutritional interventions which is a key to the recovery and reconstruction of intestinal microbiota. This is the first study in which the therapeutic effects of synbiotics are evaluated on clinical symptoms of colitis in the context of opioid. Herein, we demonstrated that modification of the gut microbiome by synbiotics can modulate the mucosal immune response and suppress the detrimental inflammation for the amelioration of IBD. Our study showed that synbiotics can protect the gut against damaging inflammation, offer the next source of IBD therapies in opioid using patients. The results of this study will help in the development of new therapeutics as “Synbiotics Therapy” for the treatment of IBD patients that are on opioids.


Talk as on 24 July,2023

Prof.Siddappa Byrareddy as a guest Speaker
Prof.Siddappa Byrareddy, University of Nebraska Medical Center (UNMC) Omaha, NE United States of America were invited for a talk at IISER Bhopal. He delivered his lecture on 24 July, 2023 at L-2, LHC, IISER BHOPAL.
Title -Immunotherapy Strategies for HIV Treatment and Prevention including CAR T cells.
Abstract-Despite the development of effective anti-retroviral therapy (ART) which effectively suppresses virus replication in the majority of HIV-infected individuals, but treatment capable of curing this infection is still not available. The gut is the first major site where HIV infection and replication take place, with CD4+ T cells that express the co-receptor CCR5 and the heterodimeric gastrointestinal tissue (GIT) homing molecule α4β7 integrin serving as the major target. Since, HIV infection is associated with a profound loss of mucosal immunological and physical integrity, which is considered a key cause of inflammation during HIV infection. Importantly, inflammation may critically contribute to HIV persistence by several mechanisms: driving the infection of susceptible cells that sustain the persistence of the reservoir; up-regulating the expression of co-inhibitory receptors, which contribute to the persistence of latently infected cells; and limiting the function of HIV-specific immune responses that could potentially clear the virus. Therefore, developing strategies aimed at limiting inflammation and improving immune responses especially in the gut and other lymphoid tissues may critically impact on HIV persistence, and is a key priority for HIV research. We have developed and tested variety of immunotherapeutic including the development of antibody therapies and more recently CAR-T cells to understand HIV/SIV reservoirs in myeloid cells in the CNS as well as gut to develop a cellular immunotherapy that targets virus-specific chimeric antigen receptor (CAR) T cells to tissue reservoirs of HIV in the CNS and lymphoid tissues to durably suppress HIV replication. In my presentation, I will be discussing some of our recent data on anti-α4β7 intervention therapy including its combination with IL- 21 supplementation and our future directions of CAR T cells to reduce SIV/HIV reservoirs.



























